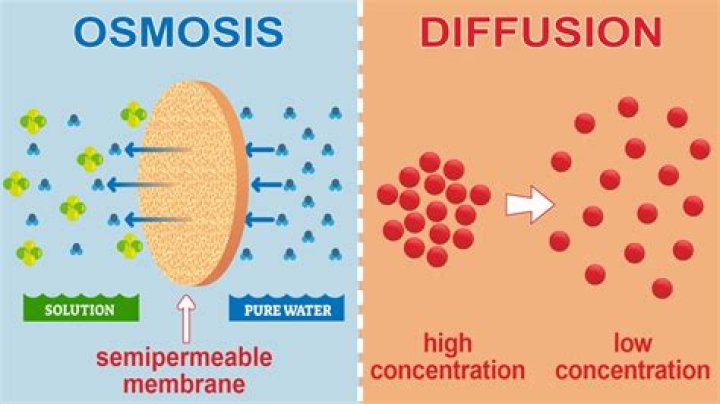
Osmosis (/?zˈmo?.s?s/) is the spontaneous net movement of solvent molecules through a selectively permeable membrane into a region of higher solute concentration, in the direction that tends to equalize the solute concentrations on the two sides. Osmosis can be made to do work..
Thereof, what happens in the process of osmosis?
It occurs when two solutions are separated by a partially permeable membrane. Osmosis is the movement of water molecules from an area of high water concentration (weak/dilute solution) to an area of low water concentration (strong/concentrated solution) through a partially permeable membrane.
Similarly, what is osmosis and example? osmosis. An example of osmosis is when red blood cells, which have a high concentration of protein and salt, are placed in a lower concentration fluid like water, the water will rush into the red blood cells.
Moreover, what is the process of osmosis and diffusion?
Diffusion is a spontaneous movement of particles from an area of high concentration to an area of low concentration. Osmosis is the spontaneous net movement of water across a semipermeable membrane from a region of low solute concentration to a more concentrated solution, up a concentration gradient.
What are the 3 types of osmosis?
The three types of osmotic conditions that affect living cells are called hypertonic, hypotonic, and isotonic states. These terms describe the osmotic state of the solution that surrounds a cell, not the solution inside the cell. Hypertonic conditions cause water to diffuse out of the cell, making the cell shrivel.
Related Question Answers
How long is osmosis?
2 to 4 hours
Is osmosis only water?
Only water or another solvent moves from a region of high energy or concentration to a region of lower energy or concentration. Diffusion can occur in any medium, whether it is liquid, solid, or gas. Osmosis occurs only in a liquid medium. Osmosis requires a semipermeable membrane.What is the process of diffusion?
Diffusion is the movement of a substance from an area of high concentration to an area of low concentration. Diffusion happens in liquids and gases because their particles move randomly from place to place. Diffusion is an important process for living things; it is how substances move in and out of cells.Why is osmosis important to humans?
The most important function of osmosis is stabilising the internal environment of an organism by keeping the water and intercellular fluids levels balanced. In all living organisms, nutrients and minerals make their way to the cells because of osmosis. This obviously is essential to the survival of a cell.What is osmosis short answer?
Osmosis is the movement of water through a semi-permeable membrane from a region of high concentration to a region of low concentration, tending to equalise the concentrations of the water. Osmosis is passive transport, meaning it does not require energy to be applied.What type of transport is osmosis?
passive transport
What causes osmosis?
Osmosis and Osmotic Pressure In microporous membranes, osmosis is caused by a momentum deficit within the pores due to the reflection of solute molecules by the membrane. This reduces the pressure on the solution side of the pore by π for a semipermeable membrane.Is osmosis a type of diffusion?
Osmosis, a type of diffusion, represents the movement of water across a partially-permeable membrane, from an area of high water concentration to an area of low water concentration.What is the opposite of diffusion?
Reverse diffusion refers to a situation where the transport of particles (atoms or molecules) in a medium occurs towards regions of higher concentration gradients, opposite to that observed during diffusion. Reverse diffusion also refers to when water is forced from a region of lower concentration to high.Is diffusion active or passive transport?
While active transport requires energy and work, passive transport does not. There are several different types of this easy movement of molecules. It could be as simple as molecules moving freely such as osmosis or diffusion. Since the cell membrane will not allow glucose to cross by diffusion, helpers are needed.What is the major difference between diffusion and osmosis?
differences: the differences between osmosis and diffusion it that diffusion refers to the movement of any chemical from one place to another, whereas osmosis exclusively refers to the movement of water across a membrane. also diffusion is the movement of molecules (solute or particles).What is water diffusion?
A diffusion is a process in science . If the particles randomly move around ("diffuse") in the water, they eventually become distributed randomly and uniformly from an area of high concentration to an area of low concentration, and organized (diffusion continues, but with no net flux).How is osmosis similar to diffusion?
Diffusion sees molecules in an area of high concentration move to areas with a lower concentration, while osmosis refers to the process by which water, or other solvents, moves through a semipermeable membrane, leaving other bits of matter in its wake.What is the best definition of osmosis?
Definition of osmosis. 1 : movement of a solvent (such as water) through a semipermeable membrane (as of a living cell) into a solution of higher solute concentration that tends to equalize the concentrations of solute on the two sides of the membrane.What is osmosis diagram?
Osmosis is the diffusion of water across a partially permeable membrane from a dilute solution (high concentration of water) to a concentrated solution (low concentration of water). In the diagram, the concentration of sugar is initially higher on the right side of the membrane.How is osmosis used in daily life?
OSMOSIS. Osmosis has a number of life-preserving functions: it assists plants in receiving water, it helps in the preservation of fruit and meat, and is even used in kidney dialysis. In addition, osmosis can be reversed to remove salt and other impurities from water.What is osmosis in water?
Mechanism. Osmosis is the movement of a solvent across a semipermeable membrane toward a higher concentration of solute (lower concentration of solvent). When a cell is submerged in water, the water molecules pass through the cell membrane from an area of low solute concentration to high solute concentration.Does osmosis occur in humans?
Osmosis occurs in both the small and large intestines, with the majority of osmosis occurring in the large intestine. As your body processes food, it moves from the esophagus to the stomach and then to the small intestine. While there, your body absorbs important nutrients via osmosis.What is osmosis learning?
“Learning through osmosis” is an analogy for natural, organic and indirect way of learning. To learn through osmosis means to learn by immersion an exposure. For example, children learn their family's native language through osmosis.