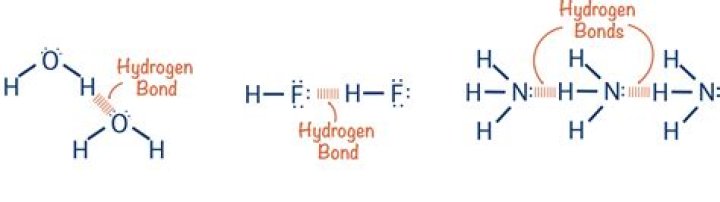
Hydrogen bonding can occur between hydrogen and four other elements. Oxygen(most common), Fluorine, Nitrogen and Carbon. Carbon is the special case in that it only really interacts in hydrogen bonding when it is bound to very electronegative elements such as Fluorine and Chlorine..
Also to know is, which elements can take part in hydrogen bonding Why is hydrogen unique in this kind of interaction?
Electronegative elements like oxygen, nitrogen, and fluorine can take part in hydrogen bonding. Hydrogen is unique in this type of bonding because the bond is formed between hydrogen atom in a polar bond and electronegative atom.
Furthermore, what else is necessary in order for a hydrogen atom in a molecule to participate in a hydrogen bond? The crystal lattice forces a less dense arrangement of molecules.
Also asked, what elements do hydrogen bond with?
Hydrogen bonding occurs only in molecules where hydrogen is covalently bonded to one of three elements: fluorine, oxygen, or nitrogen. These three elements are so electronegative that they withdraw the majority of the electron density in the covalent bond with hydrogen, leaving the H atom very electron-deficient.
Where are hydrogen bonds found?
A ubiquitous example of a hydrogen bond is found between water molecules. In a discrete water molecule, there are two hydrogen atoms and one oxygen atom.
Related Question Answers
Which hydrogen bond is strongest?
Thus, on a per bond basis, HF H bonding is strongest. However, as water has two H atoms, each molecule can form two H bonds so on a per molecule basis, water H bonding is strongest (this is evidenced by the boiling points of the three substances; NH3 < HF < H2O.)Why are hydrogen bonds important?
Hydrogen bonding is important in many chemical processes. Hydrogen bonding is responsible for water's unique solvent capabilities. Hydrogen bonds hold complementary strands of DNA together, and they are responsible for determining the three-dimensional structure of folded proteins including enzymes and antibodies.What is a hydrogen bond easy definition?
A chemical bond formed between an electropositive atom (typically hydrogen) and a strongly electronegative atom, such as oxygen or nitrogen. Hydrogen bonds are responsible for the bonding of water molecules in liquid and solid states, and are weaker than covalent and ionic bonds.How is a hydrogen bond formed?
A hydrogen bond is formed when the positive end of one molecule is attracted to the negative end of another. The concept is similar to magnetic attraction where opposite poles attract. This makes hydrogen an electrically positive atom because it has a deficiency of electrons.What is hydrogen bonding and its types?
Hydrogen bond or hydrogen bonding is a type of weak force that results in the formation of dipole-dipole interaction between a hydrogen atom and an electronegative atom that is strongly bonded to another electronegative atom. However, they are weak compared to true covalent or ionic bonds.Why are hydrogen bonds weak?
Hydrogen bonds are relatively weak, but they add to the energy needed for molecules to move apart from each other when matter changes state from a solid to a liquid or from a liquid to a gas. This explains why polar covalent compounds have relatively high melting and boiling points.Is ch4 a hydrogen bond?
Answer and Explanation: CH4 cannot form hydrogen bonds. This is because hydrogen bonds are a type of electrostatic interaction, which is only possible in molecules in whichWhat are the two conditions for the formation of hydrogen bond?
There are two requirements for hydrogen bonding. Two Requirements for Hydrogen Bonding: First molecules has hydrogen attached to a highly electronegative atom (N,O,F). Second molecule has a lone pair of electrons on a small highly electronegative atom (N,O,F).Does water have a hydrogen bond?
Hydrogen-bonding forms in liquid water as the hydrogen atoms of one water molecule are attracted towards the oxygen atom of a neighboring water molecule; generally, a proton shared by two lone electron pairs. This attraction is the basis of the 'hydrogen' bonds.How many hydrogen bonds are in A and T?
Two hydrogen bonds
Are hydrogen bonds strong or weak?
Hydrogen bonds are strong intermolecular forces created when a hydrogen atom bonded to an electronegative atom approaches a nearby electronegative atom. The hydrogen bond is one of the strongest intermolecular attractions, but weaker than a covalent or an ionic bond.How do hydrogen bonds affect physical properties?
The presence of hydrogen bonding will lift the melting and boiling points. The larger the molecule the more van der Waals attractions are possible - and those will also need more energy to break. Most molecular substances are insoluble (or only very sparingly soluble) in water.How are hydrogen bonds formed in proteins?
A hydrogen bond is mainly formed between a donor atoms such as an amine, which acts as a ligand, or a lewis base and acceptor atom like carboxylic acid. The larger the number of amino acids in a given protein molecule, the greater is the probability of the formation of hydrogen bonds.Is water polar or nonpolar?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This is an example of polar covalent chemical bonding.Which is the best description of hydrogen bonding?
Hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; such a bond is weaker than an ionic bond or covalent bond but stronger than van der Waals forces.Which of the following is an example of a hydrogen bond?
Examples of Hydrogen Bonds water (H2O): Water is an excellent example of hydrogen bonding. The bond is between the hydrogen of one water molecule and the oxygen atoms of another water molecule, not between the two hydrogen atoms (a common misconception). DNA: Hydrogen bonds form between base pairs.What type of bond is a hydrogen bond?
A hydrogen bond is the electromagnetic attraction between polar molecules in which hydrogen is bound to a larger atom, such as oxygen or nitrogen. This is not a sharing of electrons, as in a covalent bond. Instead, this is an attraction between the positive and negative poles of charged atoms.Which pairs of molecules can form a hydrogen bond with one another?
Hydrogen bond is formed only by the three highly electronegative elements- fluorine, oxygen and nitrogen. So, hydrogen bonding is possible only in those compounds in which the hydrogen atom is directly bonded to fluorine, oxygen or nitrogen.Why is water a polar molecule?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.