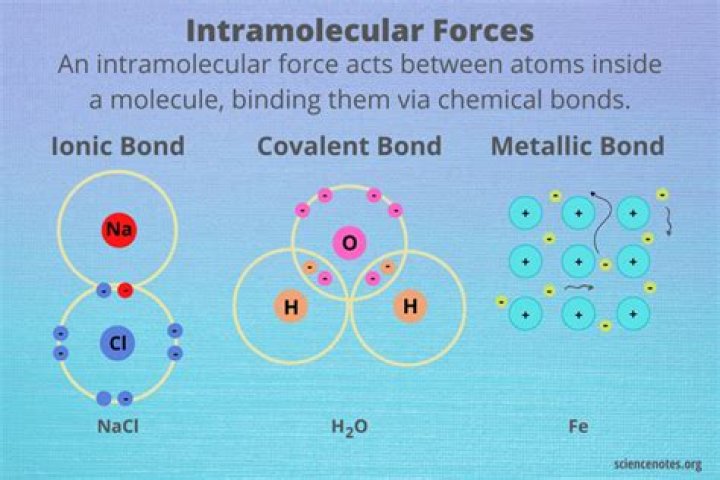
.
Also, what kind of intermolecular forces are present in br2?
ICl is polar and Br2 is nonpolar. The dominate intermolecular force in ICl is dipole- dipole whereas in Br2 it is London. Dipole-dipole is the stronger of the two and therefore more difficult to break – the melting point is higher.
Furthermore, is HCl polar or nonpolar? HCL is neither polar or non-polar. HCL is written wrong, and probably a typographical error. HCl, also known as Hydrogen Chloride is a gas at STP, and is a polar molecule. In HCl, the hydrogen atom is partially positively charged, while the chlorine atom is partially negatively charged.
Also know, does AR have dipole dipole forces?
Only one of them, NO, has a permanent electric dipole moment and so it is the only molecule which can have dipole-dipole intermolecular interactions; NO will have a higher boiling point than either Ar or CH4. The only non-vanishing intermolecular interaction in the case of Ar and CH4 is a London dispersion interaction.
Is ClF dipole dipole?
The ClF bond is a polar covalent bond and the molecule has a net dipole moment (unlike CCl4 for instance where the bond dipoles cancel giving the molecule no net dipole moment). The intermolecular interactions will be made up of dipole-dipole interactions and dispersion interactions.
Related Question AnswersIs h2s a dipole dipole?
H2S, H2Se and H2Te exhibit dipole-dipole intermolecular forces while H2O exhibits hydrogen bonding. C4H10 is a nonpolar hydrocarbon molecule so has the dispersion force (42 electrons) and has a stronger force of attraction than CO2 (bp −0.5°C).Is HCl dipole dipole?
HCl molecules, for example, have a dipole moment because the hydrogen atom has a slight positive charge and the chlorine atom has a slight negative charge. Because of the force of attraction between oppositely charged particles, there is a small dipole-dipole force of attraction between adjacent HCl molecules.Is cl2 a dipole dipole force?
H2O has dispersion, dipole-dipole, and hydrogen bonding. Cl2 has only dispersion forces and is non-polar. HCl had dispersion forces but also is dipole-dipole. SMALLER compounds are generally MORE soluble in water than larger compounds with similar structures.Is f2 dipole dipole?
Because F2 is nonpolar, they molecules aren't attracted to each other and LD IMFs result. HBr is polar, so it exhibits dipole-dipole IMFs. F2 and HBr are a pair of a polar and nonpolar molecule, so the IMFs they have are induced dipole because of the polar nature of HBr.Is ch3f polar or nonpolar?
(d) CH3F(l) – Dipole – dipole forces: CH3F is a polar molecule, it has a permanent dipole. In this case hydrogen bonding does NOT occur, since the F atom is bonded to the central C atom (F must be bonded to H in order for hydrogen bonding to occur).Is MgCl2 polar or nonpolar?
MgCl2 is polar because its molecular geometry is bent, it's not symmetrical thus leading MgCl2 being polar since the dipole forces are pulling downwards. MgCl2 is polar-ionic. This compound does have an ion-dipole force.What are the types of intermolecular forces?
The three major types of intermolecular interactions are dipole–dipole interactions, London dispersion forces (these two are often referred to collectively as van der Waals forces), and hydrogen bonds.What is the strongest intermolecular force?
hydrogen bondingHow do you measure intermolecular forces?
5. Bottom Line- Boiling points are a measure of intermolecular forces.
- The intermolecular forces increase with increasing polarization of bonds.
- The strength of intermolecular forces (and therefore impact on boiling points) is ionic > hydrogen bonding > dipole dipole > dispersion.