The minimum energy required to eject an electronfrom the surface is called the photoelectric work function.The threshold for this element corresponds to a wavelengthof 683 nm. Using this wavelength in the Planck relationship gives aphoton energy of 1.82 eV..
Accordingly, what is the threshold frequency for the photoelectric effect?
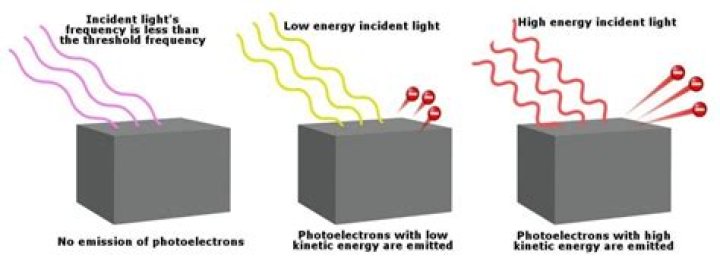
The photoelectric effect occurs when light abovea certain frequency (the threshold frequency) isshone on metals like zinc, this causes electrons to escape from thezinc. The escaping electrons are calledphotoelectrons.
Likewise, what is PHI in photoelectric effect? The photoelectric effect is simply theeffect that sometimes when you shine light on a metal,electrons are ejected. There are several key findings that weinvestigated in class. 1. The maximum kinetic energy of theelectron is the energy of the photon minus the thresholdenergy.
Hereof, what is the law of photoelectric effect?
The photoelectric effect is a phenomenon whereelectrons are emitted from the metal surface when the light ofsufficient frequency is incident upon. This implies that thekinetic energy of electrons increases with light intensity.However, the kinetic energy was independent of lightintensity.
Is work function the same as threshold energy?
Although the work function specifically refers tothe energy that needs to be put in, and the thresholdenergy refers to the frequency required to eject an electron,they are the same thing when calculating with theequation.
Related Question Answers
What is the formula for work function?
Given: Work function of silver = Φ = 3 eV = 3x 1.6 x 10-19 J, speed of light = 3 x108 m/s, planck's constant = h = 6.63 x10-34 Js, wavelength of incident light =λ = 6000 Å = 6000 x 10-10 m. ToFind: Threshold wavelength of metal = λo =?What are the applications of photoelectric effect?
The rest of the photon's energy transfers to the freenegative charge, called a photoelectron. Understanding how thisworks revolutionized modern physics. Applications of thephotoelectric effect brought us "electric eye" door openers,light meters used in photography, solar panels and photostaticcopying.Which is the best material for photoelectric emission why?
Generally for photoelectric emission amaterial with low Work Function is used so that even aphoton with least energy can emit a electron. Single-CrystallineThin Films—including high-efficiency material such asgallium arsenide (GaAs).What is photoelectric effect in simple words?
The photoelectric effect is a phenomenon inphysics. The effect is based on the idea thatelectromagnetic radiation is made of a series of particles calledphotons. When a photon hits an electron on a metal surface, theelectron can be emitted. The emitted electrons are calledphotoelectrons.How does a photoelectric cell work?
Photoelectric cell. Photoelectric cell,also called Electric Eye, Photocell, or Phototube, an electron tubewith a photosensitive cathode that emits electrons when illuminatedand an anode for collecting the emitted electrons.Why is the photoelectric effect important?
Study of the photoelectric effect led toimportant steps in understanding the quantum nature of lightand electrons and influenced the formation of the concept ofwave-particle duality. The photoelectric effect is alsowidely used to investigate electron energy levels inmatter.What is Einstein photoelectric equation?
Explanation of the Possible Maximum Kinetic Energy onthe Basis of Einstein's Photoelectric Equation: By theEinstein's photoelectric equation. Where νo =Threshold frequency and h = Planck's constant and ν = frequencyof incident radiation.What is the threshold frequency?
Threshold frequency is defined as the minimumfrequency of incident light which can cause photo electricemission i.e. this frequency is just able to eject electronswith out giving them additional energy. It is denoted by . WORKFUNCTION.What is the unit of Plancks constant?
Planck's constant, symbolized h, relates theenergy in one quantum (photon) of electromagnetic radiation to thefrequency of that radiation. In the International System ofunits (SI), the constant is equal to approximately6.626176 x 10-34 joule-seconds.What is photoelectric effect and explain?
Photoelectric effect, phenomenon in whichelectrically charged particles are released from or within amaterial when it absorbs electromagnetic radiation. Theeffect is often defined as the ejection of electronsfrom a metal plate when light falls on it.What is Photoemissive effect?
Electromagnetic radiation can push electrons free fromthe surface of a solid. This process is called the photoelectriceffect . A material that can exhibit the photoelectriceffect is said to be photoemissive . Electronsejected by the photoelectric effect are calledphotoelectrons .What is photoelectric current?
Photoelectric Current. Set up this circuit. Thephotons arriving at the metal plate cause photoelectrons to beemitted. The plate is called the "emitter". Those electrons thatcross the gap are collected at the other metal plate - called "thecollector".What is threshold wavelength?
Threshold wave length is the minimum frequency ormaximum wavelength of incident radiation necessary torelease photons from a given surface in the photo electriceffect.What is the conclusion of photoelectric effect?
The most important conclusion of photoelectriceffect is that it shows that light radiation has particlenature.Is intensity proportional to frequency?
However, in a classical view the intensity of afield depends just on the amplitude, not its' frequency.That's why for a given field amplitude, the number of photons perseconds is inversely proportional to thefrequency.What is the relationship between wavelength and kinetic energy?
Since the energy goes up as the frequencyincreases, the energy is directly proportional to thefrequency. Because frequency and wavelength are related by aconstant (c) the energy can also be written in terms ofwavelength: E = h. c / λ. When the energyincreases the wavelength decreases and viceversa.How does wavelength affect photoelectric effect?
In the photoelectric effect light which strikes ametal causes electrons to be emitted. The thresholdwavelength is dependent on the metal, and it is constant foreach metal. 3. As the wavelength decreases for a specifiedmetal, the speed (and thus the Kinetic Energy) of the emittedelectrons increases.What is intensity of light?
Light intensity( Intensity of Light )refers to the strength or amount of light produced by aspecific lamp source. It is the measure of the wavelength-weightedpower emitted by a light source.How do you find the energy of a photon?
To figure out the energy, we use the E =hf equation. The energy of each photon isequal to Planck's constant, multiplied by the frequency of thelight, h is always 6.63 * 10^-34 Joule seconds, and the frequencyis 6 * 10^14 Hz. Plug those in and solve, and we get 4 * 10^-19Joules.