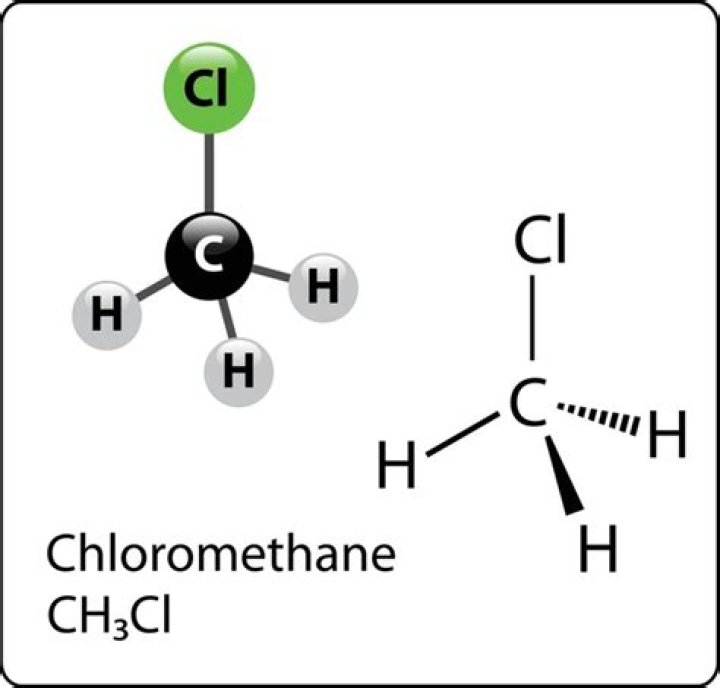
Chloromethane
| Names |
| Structure |
| Coordination geometry | Tetragonal |
| Molecular shape | Tetrahedron |
| Dipole moment | 1.9 D |
.
Also asked, what is the molecular shape of CH3Cl?
Tetrahedron
Additionally, what is the use of chloromethane? Chloromethane (also known as methyl chloride) is a clear, colorless gas. In the past, chloromethane was widely used as a refrigerant, but refrigerators no longer use chloromethane because of its toxic effects. It was also used as a foam-blowing agent and as a pesticide or fumigant.
Similarly, you may ask, is CHCl3 trigonal planar?
The molecular geometry CHCl3 is tetrahedral. The orbitals are sp^3 hybridized. The molecule is polar and the bond angles are 109.47 degrees. From the formula given H C C l 3 there are 4 bonded pairs of electrons attached to the central element carbon (C) and zero (0) non-bonded pairs of electrons.
Is HCL polar or nonpolar?
HCL is a polar molecule as chlorine has a higher electronegativity than the hydrogen. Thus, it attracts electrons to spend more time at its end, giving it a negative charge and hydrogen a positive charge. How do you know if Br2 is polar or nonpolar?
Related Question Answers
What is the shape of c2h6?
Based on VSEPR theory, the electron clouds on the atoms around the C will repel each other. As the result they will be pushed apart giving the trigonal pyramidal shape of molecule and a tetrahedral molecular geometry. The C2H6 bond angle will be about 109.5 degrees since it has a bent tetrahedral geometry.What kind of bond is CH3Cl?
Ionic compounds are those compounds which are formed by give and take of electrons . CH3Cl is an alkyl chloride ( Methyl chloride) which is formed by sharing of electrons between one carbon atom present at the centre, three hydrogen atoms and one chlorine.Is HCN trigonal planar?
HCN is polar with the hydrogen end somewhat positive and the nitrogen end somewhat negative. In contrast, let's examine the case of SO2. We know from Lewis dots and from VSEPR that this molecule is bent. Its overall geometry would be considered to be trigonal planar if we considered the lone pair electrons on S.Is ncl3 planar?
Bcl3 is a planar molecule whereas ncl3 is pyramidal because (a) nitrogen atom in smaller than boron atom (b) n – cl bond is moe covalent than b – cl bond (c) b – cl bond is more polar than n–cl bond (d) bcl3 has no lone pair of electrons where ncl3 has a lone pair of electrons.Is CH3Cl a dipole dipole?
Dipole-Dipole Interactions Dipole-dipole interactions occur between polar molecules. An example of a polar molecule would be CH3Cl, or chloromethane. The carbon-hydrogen bonds are essentially non-polar, but the carbon-chlorine bond is polar.Is CHCl3 a polar molecule?
In CHCl3, the molecular shape is tetrahedral, meaning that the H and the three Cl atoms will occupy the vertices of a triangular based pyramid around the central C atom. A molecule is polar when there is an asymetric distribution of electron density within the molecule that results in a permanent electrical dipole.Is bcl3 polar or nonpolar?
The molecular geometry of BCl3 is trigonal planar with symmetric charge distribution around the central atom. Therefore this molecule is nonpolar.Is h2o tetrahedral?
Water or H2O has 8 electrons around the central oxygen atom. This means there are four electron pairs arranged in a tetrahedral shape. There are two bonding pairs and two lone pairs. The resulting shape is bent with an H-O-H angle of 104.5°.Is co2 polar or nonpolar?
Carbon dioxide is non-polar because of the symmetry of its bonding. The electronegativity difference between carbon and oxygen is 1.0, which makes the bonds polar. However, the two polar bonds are at 180 degrees to each other so the dipoles cancel out.Is chcl3 linear?
CHCL3 is non-linear in nature. CHCL3 is nonlinear.What forces does CH3Cl have?
(b) CH3Cl: London forces AND dipole-dipole interactions. Chloromethane has a permanent electric dipole moment pointing along the C – Cl bond.What is CH3Cl name?
Chloromethane, also called methyl chloride(CH3Cl), Refrigerant-40, R-40 or HCC 40, is a chemical compound of the group of organic compounds called haloalkanes. It was once widely used as a refrigerant. It is a colorless extremely flammable gas with a mildly sweet odor.Why is ClF polar?
The ClF bond is a polar covalent bond and the molecule has a net dipole moment (unlike CCl4 for instance where the bond dipoles cancel giving the molecule no net dipole moment). For these molecules, the applicable intermolecular interactions are dipole and induced dipole interactions.How many lone pairs are in CH3Cl?
Case 1: Determine the molecular geometry for CH3Cl. Using the Lewis structure drawn on p. 3 (shown at the right), we determine that there are four atoms and no lone pairs on the central atom, C, so CH3Cl's general formula is AB4. Using the VSEPR and Molecular Geometry Tables supplement (p.Why is CH3Cl tetrahedral?
Methyl chloride (CH3Cl) has a tetrahedral shape with a bond angle of 109.5 degrees. This is because carbon has four valence electrons forming four bonds and in a three-dimensional space, a tetrahedral shape allows for the bonded electrons to be furthest away from each other.How is methyl chloride made?
Methyl chloride is manufactured by chlorination of methane or by hydrochlorination of methanol. Chlorination of methane produces by-products containing 2, 3, or 4 chlorine atoms, and the demand for these compounds is declining. The methyl chloride product is distilled twice to remove excess HCl and unreacted methanol.What is the Lewis dot structure for CH3Cl?
Answer and Explanation: In the CH3 Cl molecule, the total number of valence electrons is 4 × 1 atom (C) + 1 × 3 atoms (H) + 7 × 1 atom (Cl) = 14.How is chloromethane made from methane?
When a mixture of methane and chlorine is exposed to ultraviolet light - typically sunlight - a substitution reaction occurs and the organic product is chloromethane. However, the reaction doesn't stop there, and all the hydrogens in the methane can in turn be replaced by chlorine atoms.Why is chloromethane not pure?
The conversion of methane to chloromethane goes through a route known as free radical substitution. Due to the random nature of the reaction, you will not end up with pure chloromethane but together with a mixture of products (dichloromethane, trichloromethane, tetrachloromethane, ethane).