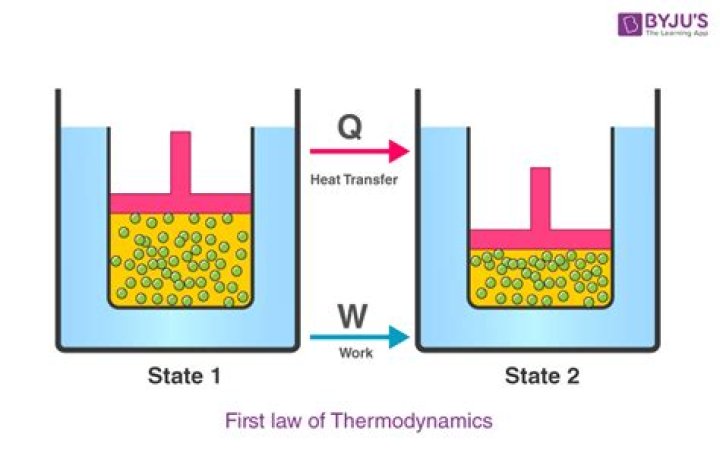
Since the First Law of Thermodynamics states that energy is not created nor destroyed we know that anything lost by the surroundings is gained by the system. Therefore, q and w are positive in the equation ΔU=q+w because the system gains heat and gets work done on itself..
In this regard, what is an example of the 1st Law of Thermodynamics?
Examples of the First Law of Thermodynamics. Energy Flow in a Diesel Engine. When an engine burns fuel it converts the energy stored in the fuel's chemical bonds into useful mechanical work and into heat.
Additionally, what is the first law of thermodynamics and how does it relate to energy use? The first law of thermodynamics states that energy is conserved in chemical processes. In every process that involves energy, some of the energy is lost to the surroundings as heat. This means that no process is perfectly efficient.
In this regard, what is second law of thermodynamics in chemistry?
The Second Law of Thermodynamics states that the state of entropy of the entire universe, as an isolated system, will always increase over time. The second law also states that the changes in the entropy in the universe can never be negative.
What does the 2nd law of thermodynamics say?
The Second Law of Thermodynamics says that processes that involve the transfer or conversion of heat energy are irreversible. The First Law of Thermodynamics states that energy cannot be created or destroyed; the total quantity of energy in the universe stays the same.
Related Question Answers
How do you define enthalpy?
Enthalpy is a thermodynamic property of a system. It is the sum of the internal energy added to the product of the pressure and volume of the system. It reflects the capacity to do non-mechanical work and the capacity to release heat. Enthalpy is denoted as H; specific enthalpy denoted as h.What is the application of First Law of Thermodynamics?
The first law of thermodynamics is the application of the conservation of energy principle to heat and thermodynamic processes: The first law makes use of the key concepts of internal energy, heat, and system work. It is used extensively in the discussion of heat engines.Why is First Law of Thermodynamics important?
The first law of thermodynamics, arguably the most important, is an expression of the principle of conservation of energy. Consistent with this principle, the first law expresses that energy can be transformed (i.e. changed from one form to another), but cannot be created or destroyed.What are the examples of thermodynamics?
These laws are observed regularly every day. - Melting Ice Cube. Every day, ice needs to be maintained at a temperature below the freezing point of water to remain solid.
- Sweating in a Crowded Room. The human body obeys the laws of thermodynamics.
- Taking a Bath.
- Flipping a Light Switch.
What are the 1st 2nd and 3rd laws of thermodynamics?
The first law, also known as Law of Conservation of Energy, states that energy cannot be created or destroyed in an isolated system. The second law of thermodynamics states that the entropy of any isolated system always increases.What is first law of thermodynamics in chemistry?
Chemistry Glossary Definition of First Law of Thermodynamics The first law of thermodynamics is the physical law which states that the total energy of a system and its surroundings remain constant.What are the first three laws of thermodynamics?
The three laws of thermodynamics are: First law of thermodynamics: When energy passes, as work, as heat, or with matter, into or out of a system, the system's internal energy changes in accord with the law of conservation of energy.Who made the first law of thermodynamics?
Rudolf Clausius
What is the unit of entropy?
The SI unit for Entropy (S) is Joules per Kelvin (J/K). A more positive value of entropy means a reaction is more likely to happen spontaneously.What is the opposite of entropy?
Negentropy is reverse entropy. It means things becoming more in order. By 'order' is meant organisation, structure and function: the opposite of randomness or chaos. One example of negentropy is a star system such as the Solar System. The opposite of entropy is negentropy.Why is entropy important?
Entropy is a bookkeeping device, which tells us about the flow and distribution of energy. For any process to occur to occur spontaneously, it is a necessary condition that the entropy of the system undergoing the process should increase. If the entropy decreases, then that process cannot occur spontaneously.What are the 4 laws of thermodynamics?
The 4 Laws First law of thermodynamics – Energy can neither be created nor destroyed. It can only change forms. In any process, the total energy of the universe remains the same. Third law of thermodynamics – As temperature approaches absolute zero, the entropy of a system approaches a constant minimum.Why is second law of thermodynamics important?
Second law of thermodynamics is very important because it talks about entropy and as we have discussed, 'entropy dictates whether or not a process or a reaction is going to be spontaneous'.What is Newton's second law?
Newton's second law of motion pertains to the behavior of objects for which all existing forces are not balanced. The second law states that the acceleration of an object is dependent upon two variables - the net force acting upon the object and the mass of the object.Which has the highest entropy?
Solids have the fewest microstates and thus the lowest entropy. Liquids have more microstates (since the molecules can translate) and thus have a higher entropy. When a substance is a gas it has many more microstates and thus have the highest entropy.How do I calculate entropy?
But entropy change is quoted in energy units of J. That means that if you are calculating entropy change, you must multiply the enthalpy change value by 1000. So if, say, you have an enthalpy change of -92.2 kJ mol-1, the value you must put into the equation is -92200 J mol-1.What is entropy in chemistry?
In chemistry, entropy is represented by the capital letter S, and it is a thermodynamic function that describes the randomness and disorder of molecules based on the number of different arrangements available to them in a given system or reaction.What is the importance of thermodynamics?
Law of Thermodynamics Thermodynamics is a very important branch of both physics and chemistry. It deals with the study of energy, the conversion of energy between different forms and the ability of energy to do work.What is entropy in biology?
Entropy, the measure of a system's thermal energy per unit temperature that is unavailable for doing useful work. Because work is obtained from ordered molecular motion, the amount of entropy is also a measure of the molecular disorder, or randomness, of a system.