.
Also question is, what is activation energy in chemistry?
Activation energy, in chemistry, the minimum amount of energy that is required to activate atoms or molecules to a condition in which they can undergo chemical transformation or physical transport.
Likewise, what is an example of activation energy? This energy is called activation energy. For example, activation energy is needed to start a car engine. Turning the key causes a spark that activates the burning of gasoline in the engine. The combustion of gas won't occur without the spark of energy to begin the reaction.
Besides, what is the best definition of activation energy?
the energy required to end a chemical reaction. the energy required to bind a substrate to an active site. the energy required to break the bonds of reactant molecules.
What kind of energy is activation energy?
In chemistry and physics, Energy of Activation is the energy which must be provided to a chemical or nuclear system with potential reactants to result in: a chemical reaction, nuclear reaction, or various other physical phenomena.
Related Question AnswersHow do you explain activation energy?
Threshold energy is the total amount of energy the reactants need to attain the transition state. Activation energy is the added extra energy (generally kinetic) that molecules need to reach the threshold energy. I.e. it is the difference between the average internal energy of the reactants and the threshold energy.What do u mean by activation energy?
Activation Energy. The term Activation Energy was introduced in 1889 by Svante Arrhenius, a Swedish scientist. It is defined as the least possible amount of energy (minimum) which is required to start a reaction or the amount of energy available in a chemical system for a reaction to take place.What factors affect activation energy?
Reactions occur when two reactant molecules effectively collide, each having minimum energy and correct orientation. Reactant concentration, the physical state of the reactants, and surface area, temperature, and the presence of a catalyst are the four main factors that affect reaction rate.Who discovered activation energy?
Svante ArrheniusIs activation energy positive or negative?
Originally Answered: is activation energy negative? Activation energy is typically positive. It's the minimum energy required for a chemical reaction to proceed. There are few other ways to describe the activation energy but they basically say the same thing.Why is activation energy important?
All chemical reactions, including exothermic reactions, need activation energy to get started. Activation energy is needed so reactants can move together, overcome forces of repulsion, and start breaking bonds.What is the role of activation energy?
Activation energy is defined as the minimum amount of energy required by the reactant molecules to undergo a chemical reaction. But when we add a catalyst into the reaction then there will be a decrease in activation energy and as a result, molecules will lesser amount of energy are able to participate in the reaction.Is activation energy the same as enthalpy?
For reactants to form products in an reaction, an initial amount of energy i.e. activation energy needs to be provided. This is called activation energy. The enthalpy of reaction, is simply the enthalpy of products minus the enthalpy of reactants.What do you mean by free energy?
In physics and physical chemistry, free energy refers to the amount of internal energy of a thermodynamic system that is available to perform work. Helmholtz free energy is energy that may be converted into work at constant temperature and volume.What is an example of a catalyst?
Chemical Catalysts Hydrogen peroxide will decompose into water and oxygen gas. Two molecules of hydrogen peroxide will produce two molecules of water and one molecule of oxygen. A catalyst of potassium permanganate can be used to speed up this process.How do you find the activation energy for the reverse reaction?
- ln(50) = (30)e-Ea/(8.314)(679)
- Ea = 11500 J/mol.
- Because the reverse reaction's activation energy is the activation energy of the forward reaction plus ΔH of the reaction:
- 11500 J/mol + (23 kJ/mol X 1000) = 34500 J/mol.
What is the definition of activation energy in biology?
In biology activation energy is defines as the energy required to start a reaction. Enzymes are proteins that bind to a molecule, or substrate, to modify it and lower the energy required to make it react.What do you mean by enzymes?
Enzyme: Proteins that speeds up the rate of a chemical reaction in a living organism. An enzyme acts as catalyst for specific chemical reactions, converting a specific set of reactants (called substrates) into specific products. Without enzymes, life as we know it would not exist.What is activation energy simple?
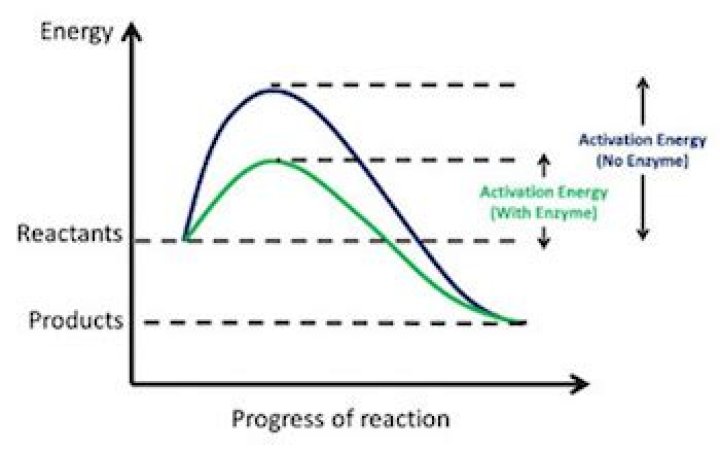
The activation energy of a chemical reaction is the minimum energy that is needed to make the reaction happen. It can be thought of as a barrier between the reagents and the products of a reaction. The activation energy is the difference in energy between the transition state and the starting reagents.What is the activation energy of an enzyme?
The activation energy is the energy required to start a reaction. Enzymes are proteins that bind to a molecule, or substrate, to modify it and lower the energy required to make it react. The rate of reaction is given by the Arrhenius equation.What is a catalyst choose the best definition?
PLAY. Match. The best definition of catalysis is: (Choose the ONE best answer) making a reaction more likely to proceed.What is an energy diagram for a chemical reaction?
Potential Energy Diagrams. The energy changes that occur during a chemical reaction can be shown in a diagram called a potential energy diagram, or sometimes called a reaction progress curve. A potential energy diagram shows the change in potential energy of a system as reactants are converted into products.Is ATP an enzyme?
ATP synthase is an enzyme that creates the energy storage molecule adenosine triphosphate (ATP). ATP is the most commonly used "energy currency" of cells for all organisms.How do you draw an activation energy diagram?
1 Answer- Draw and label a pair of axes. Label the vertical axis "Potential Energy" and the horizontal axis "Reaction Coordinate".
- Draw and label two short horizontal lines to mark the energies of the reactants and products.
- Draw the energy level diagram.
- Draw and label the activation energy.