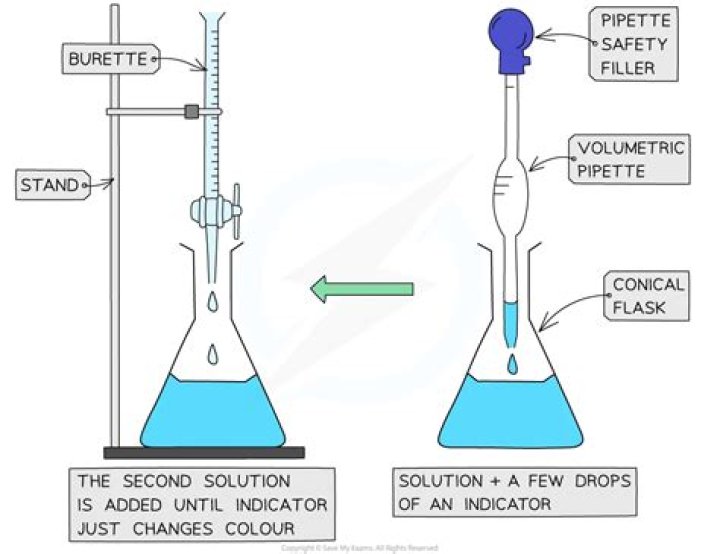
The apparatus used in a titration are burette, pipette(most of the times 10 ml ones are used), measuring flask(250 ml), two beakers (100ml and 250 ml), measuring cylinder( used while taking 10 ml 4N H2SO4) and a burette stand..
In this regard, how is titration used in industry?
Titration is an analytical technique that is widely used in the food industry. It allows food manufacturers to determine the quantity of a reactant in a sample. For example, it can be used to discover the amount of salt or sugar in a product or the concentration of vitamin C or E, which has an effect on product colour.
what are redox titrations used for in industry? The titrant is the standardized solution; the analyte is the analyzed substance. Redox titration determines the concentration of an unknown solution (analyte) that contains an oxidizing or reducing agent. Not all titrations require an external indicator.
Moreover, what is the name of the piece of equipment used for titration analysis?
Acid-base titrations are used to determine the concentration of a sample of acid or base and are carried out using a piece of equipment called a burette. It is a long, glass tube with a tap at the end which can be used to very carefully add drops of liquid to a test solution.
What is the purpose of titration?
The concentration of a basic solution can be determined by titrating it with a volume of a standard acid solution (of known concentration) required to neutralize it. The purpose of the titration is the detection of the equivalence point, the point at which chemically equivalent amounts of the reactants have been mixed.
Related Question Answers
How are titrations used in real life?
Many real life uses of titration are used each day, especially in the medical world and labs. Pharmacists use it to get the proper mix when compounding medicines. It's used to get the necessary proportions in intravenous drips. Titration is used to measure blood sugar levels and to analyze blood for other diagnoses.What is different type of titration?
Types of Titrations • Acid-base titrations, in which an acidic or basic titrant reacts with an analyte that is a base or an acid. Precipitation titrations, in which the analyte and titrant react to form a precipitate. • Redox titrations, where the titrant is an oxidizing or reducing agent.Where is volumetric analysis used in industry?
Volumetric analysis and titration are in wide use in a variety of industries because they are considered a basic technique in analytical chemistry. For example, titration can be used by the biodiesel industry to determine the acidity of a sample of vegetable oil.How is titration used in the waste oil industry?
Titration is an indirect test for free fatty acids (FFA) in waste restaurant fryer oil. Titration is done by reacting a small sample of free fatty acid (which happens to be in a ml of waste oil) with a measured amount of lye, and using pH to tell us when the FFA is all used up.How is titration used in pharmacology?
Drug titration is the process of adjusting the dose of a medication for the maximum benefit without adverse effects. Drug titration is also used in phase I of clinical trials. The experimental drug is given in increasing dosages until side effects become intolerable.Why is accuracy important in titration?
Titrations are carried out using the concentrations that we do for very good reasons: The end point is well defined and the accuracy is good. As you increase the concentration the accuracy decreases, there is more chance of 'overshooting' the endpoint.How titration is used in medicine?
Titration is used in laboratory medicine to determine unknown concentrations of chemicals of interest in blood and urine. Titration may also be used to determine the amount of a certain chemical in food. Often, titration is used to determine fat content, water content, and concentrations of vitamins.What is the formula for titration?
Use the titration formula. If the titrant and analyte have a 1:1 mole ratio, the formula is molarity (M) of the acid x volume (V) of the acid = molarity (M) of the base x volume (V) of the base. (Molarity is the concentration of a solution expressed as the number of moles of solute per litre of solution.)What is the process of titration?
A titration is a technique where a solution of known concentration is used to determine the concentration of an unknown solution. Typically, the titrant (the know solution) is added from a buret to a known quantity of the analyte (the unknown solution) until the reaction is complete.What is end point in titration?
End Point. end point: the point during a titration when an indicator shows that the amount of reactant necessary for a complete reaction has been added to a solution.Is NaOH an acid or base?
NaOH is a base because when dissolved in water it dissociates into Na+ and OH- ions. It is the OH- (hydroxyl ion) which makes NaOH a base. In classical term a base is defined as a compound which reacts with an acid to form salt and water as depicted by the following equation. NaOH+HCl=NaCl+H2O.What is indicator in titration?
Indicator: A substance that changes color in response to a chemical change. An acid–base indicator (e.g., phenolphthalein) changes color depending on the pH. Redox indicators are also used. A drop of indicator solution is added to the titration at the beginning; the endpoint has been reached when the color changes.What is the pH of an indicator?
pH indicators are weak acids that exist as natural dyes and indicate the concentration of H+ (H3O+) ions in a solution via color change. A pH value is determined from the negative logarithm of this concentration and is used to indicate the acidic, basic, or neutral character of the substance you are testing.How do indicators work?
HOW DO pH INDICATORS WORK? pH indicators detect the presence of H+ and OH-. They do this by reacting with H+ and OH-: they are themselves weak acids and bases. If an indicator is a weak acid and is coloured and its conjugate base has a different colour, deprotonation causes a colour change.What type of reaction is an acid base titration?
An acid – base titration is used to determine the unknown concentration of an acid or base by neutralizing it with an acid or base of known concentration. Neutralization is the reaction between an acid and a base, producing a salt and a neutralized base.Why KMnO4 is a self indicator?
So once all the permanganate ions are used up in the reaction, the solution loses its pink colour. This indicates the end of the reaction and hence potassium permanganate is called a self indicator as it acts as an indicator apart from being one of the reactants.Why KMnO4 is widely used in redox titrations?
The oxalic acid acts as a reducing agent, and the KMnO4 acts as an oxidizing agent. KMnO4 acts as an indicator of where the permanganate ions are a deep purple colour. In this redox titration, MnO4– is reduced to colourless manganous ions (Mn2+) in the acidic medium.Why is KMnO4 used in titration?
Potassium Permanganate is an oxidizing agent, which is of deep violet colour. When used in redox titration, it get reduced into brown coloured Mn2+ ion(In acidic media) at end point and colour change at end point can be detected easily.What is the principle of redox titration?
PRINCIPLE. The principle involved in the oxidation-reduction titrations is that the oxidation process involves the loss of electrons whereas the reduction process involves the gain of electrons.