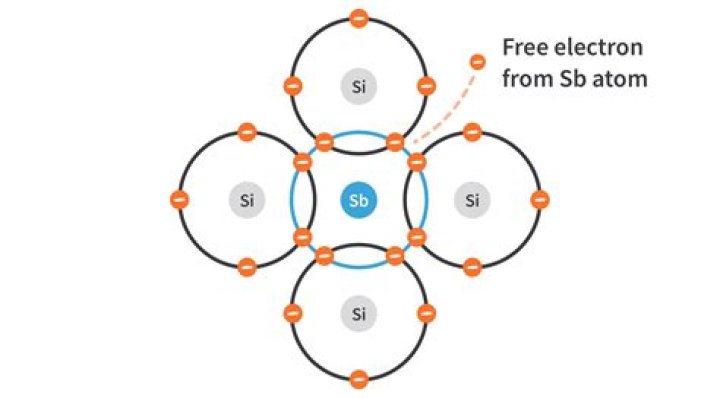
Pentavalent elements are those elements which have five electrons in their outer shell. To make the n-type semiconductor, pentavalent impurities like phosphorus or arsenic are added. Four of the impurities' electrons form bonds with the surrounding silicon atoms. This leaves one electron free..
Keeping this in consideration, what are trivalent elements?
Trivalent impurities. Trivalent impurity atoms have 3 valence electrons. The various examples of trivalent impurities include Boron (B), Gallium (G), Indium(In), Aluminium(Al). Boron is a substance consisting of atoms which all have the same number of protons. The atomic number of boron is 5 i.e. 5 protons.
Similarly, how N type material is formed? An N - type semiconductor is formed when a small amount of pentavalent impurity is added to a pure Germenium or Silicon crystal. The addition of pentavalent impurity produces a large no. of free electrons in the host crystal.
Beside this, what are trivalent and pentavalent impurities?
trivalent impurities atom has 3 valence electrons. whereas. group 15 in periodic table consist of pentavalent impurities. pentavelant impurities atom has 5 valence electrons.
What is n type and p type material?
In n-type semiconductors, electrons are the majority carriers and holes are the minority carriers. P-type semiconductors are created by doping an intrinsic semiconductor with acceptor impurities (or doping an n-type semiconductor). A common p-type dopant for silicon is boron.
Related Question Answers
What is an impurity element?
An impurity in the crystal is an atom of another element located either in place of one of the crystal's atoms or in the spaces between crystal atoms. If the impurity is located in place of one of the semiconductor atoms we call it a dopant.What is a band in chemistry?
Definition of Solid State Band Theory Because of the very large number of atoms that interact in a solid material, the energy levels are so closely spaced that they form bands. The highest energy filled band, which is analogous to the highest occupied molecular orbital in a molecule (HOMO), is called the valence band.What is Group 13 on the periodic table?
Group 13 is sometimes referred to as the boron group, named for the first element in the family. These elements are--not surprisingly--located in column 13 of the periodic table. This group includes boron, aluminum, gallium, indium, thallium, and ununtrium (B, Al, Ga, In, Tl, and Uut, respectively).What is the difference between N and P type semiconductors?
Difference Between p-Type and n-Type Semiconductor. In a p-type semiconductor, the III group element of the periodic table is added as a doping element, whereas in n-type the V group element is the doping element. In a p-type semiconductor, the majority carriers are holes, and minority carriers are electrons.What is the difference between intrinsic and extrinsic semiconductors?
The difference between intrinsic and extrinsic semiconductor is that Intrinsic semiconductors are the pure form of semiconductor materials. Whereas extrinsic semiconductors are impure semiconductor formed by adding an impurity to a pure semiconductor.What is acceptor impurity?
An acceptor Impurity is a physical material which when added to a semiconductor can form P-type region by creating positive charges or holes in the semiconductor material like silicon or germanium. Log In|Register. Shopping Cart >What are the two types of dopants?
Silicon dopants - Phosphorus is a n-type dopant.
- Arsenic is a n-type dopant.
- Antimony is a n-type dopant.
- Bismuth is a promising dopant for long-wavelength infrared photoconduction silicon detectors, a viable n-type alternative to the p-type gallium-doped material.
How semiconductors are formed?
Semiconductors are made from materials that have free electrons in their structure that can move easily between atoms, which aids the flow of electricity. Silicon has four electrons in its outer orbital, which allows the covalent bonds to form a lattice and thus form a crystal.What is P type impurities?
Pentavalent impurities Impurity atoms with 5 valence electrons produce n-type semiconductors by contributing extra electrons. Trivalent impurities Impurity atoms with 3 valence electrons produce p-type semiconductors by producing a "hole" or electron deficiency.What is forbidden gap?
Answer: When atoms are brought close together, as in a solid, the electrons come under the influence of forces from other atoms, where the energy level merges into bands of energy levels. Separating these two bands is an energy gap , termed the forbidden gap, in which electrons cannot normally exist.Why impurities are added to semiconductor?
The conductivity of semiconductors may easily be modified by introducing impurities into their crystal lattice. The process of adding controlled impurities to a semiconductor is known as doping. The amount of impurity, or dopant, added to an intrinsic (pure) semiconductor varies its level of conductivity.What are pentavalent impurities?
Pentavalent elements are those elements which have five electrons in their outer shell. To make the n-type semiconductor, pentavalent impurities like phosphorus or arsenic are added. Four of the impurities' electrons form bonds with the surrounding silicon atoms. This leaves one electron free.What is the meaning of pentavalent impurity?
Since pentavalent atom i.e dopant having valency 5 i.e, an element whose atom has 5 valence electrons is called pentavalent impurity. For example. As Pb, phosphorous, etc. These impurities are known as Donor impurities i.e. because they donate extra free electrons to the intrinsic semiconductor.Why Germanium is preferred over the silicon?
The structure of Germanium crystals will be destroyed at higher temperature. However, Silicon crystals are not easily damaged by excess heat. Peak Inverse Voltage ratings of Silicon diodes are greater than Germanium diodes. Si is less expensive due to the greater abundance of element.How many types of semiconductors are there?
two
Why impurity doped silicon is a semiconductor?
Impurity doped silicon is a semiconductor. In case of boron : boron form three bonds with silicon, it will result in an electron deficient bond andwill create a hole. These holes can move through crystal like positive charge giving rise electrical conductivity.What are acceptor and donor impurities?
In the previous sections it was considered that two types of impurities are used for doping semiconductors: donor impurities and acceptor impurities. A semiconductor that is doped with a donor impurity is called an n-type semiconductor. One that is doped with an acceptor impurity is called a p-type semiconductor.What is an N type?
A N-type semiconductor is defined as a type of extrinsic semiconductor doped with a pentavalent impurity element which has five electrons in its valence shell. The pentavalent impurity or dopant elements are added in the N-type semiconductor to increase the number of electrons for conduction.What is N type semiconductor with example?
N-type Semiconductor Example A N-type semiconductor is created by doping this pure silicon crystal lattice with a pentavalent impurity element like Antimony (Sb). The fifth electron of the impurity atom is not bonded with any semiconductor atom in the crystal lattice.