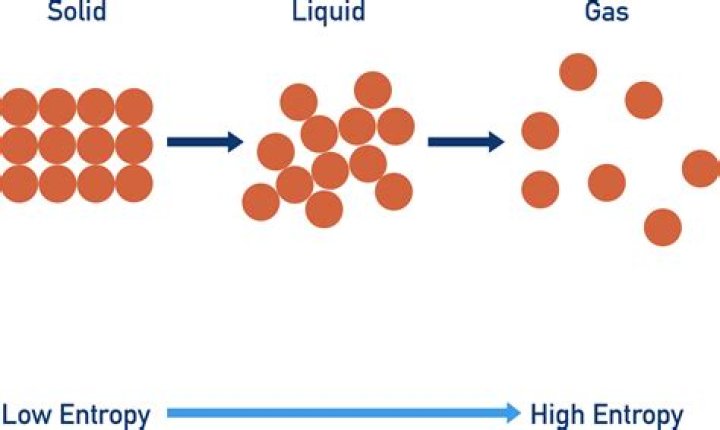
As the current knowledge of science is (the laws ofthermodynamic), the entropy of a closed system cannot bereversed. Entropy is not a process, but a a quantitythat measures how "random" or "disordered" a system is. The SecondLaw of Thermodynamics says that entropy only ever increasesspontaneously..
In this manner, can entropy be decreased?
It just says that the total entropy of theuniverse can never decrease. Entropy candecrease somewhere, provided it increases somewhere else by atleast as much. The entropy of a system decreases onlywhen it interacts with some other system whose entropyincreases in the process. That is the law.
what is the opposite of entropy? Negentropy is reverse entropy. It means thingsbecoming more in order. By 'order' is meant organisation, structureand function: the opposite of randomness or chaos. Oneexample of negentropy is a star system such as the Solar System.The opposite of entropy is negentropy.
Besides, is the entropy of the universe increasing or decreasing?
This statement is usually called the principle ofentropy increase. Irreversible or spontaneous processes canoccur only in that? direction for which the entropy of theuniverse or that of an isolated system, increases. Theseprocesses cannot occur in the direction of decreasingentropy.
Can entropy decrease in a closed system?
In a closed system, available energy cannever increase, so (because energy is conserved) its complement,entropy, can never decrease. A familiardemonstration of the second law is the flow of heat from hot thingsto cold, and never vice-versa. During this process, theentropy of the system increases.
Related Question Answers
What happens when entropy increases?
Affecting Entropy If you increase temperature, you increaseentropy. (1) More energy put into a system excites themolecules and the amount of random activity. (2) As a gas expandsin a system, entropy increases. (3) When a solid becomes aliquid, its entropy increases.What is the formula for entropy?
Entropy has the dimension of energy divided bytemperature, which has a unit of joules per kelvin (J/K) in theInternational System of Units.What are the three laws of thermodynamics?
The three laws of thermodynamics define physicalquantities (temperature, energy, and entropy) that characterizethermodynamic systems at thermal equilibrium. Third lawof thermodynamics: The entropy of a system approaches aconstant value as the temperature approaches absolutezero.What does Second Law of Thermodynamics mean?
The First Law of Thermodynamics states thatenergy cannot be created or destroyed; the total quantity of energyin the universe stays the same. The Second Law ofThermodynamics is about the quality of energy. It states thatas energy is transferred or transformed, more and more of it iswasted.Why is entropy important in thermodynamics?
It is in this sense that entropy is a measure ofthe energy in a system that cannot be used to do work. Anirreversible process degrades the performance of athermodynamic system, designed to do work or producecooling, and results in entropy production. Theentropy generation during a reversible process iszero.Does entropy mean disorder?
A measure of the unavailability of a system's energy todo work; also a measure of disorder; the higher theentropy the greater the disorder. In thermodynamics,a parameter representing the state of disorder of a systemat the atomic, ionic, or molecular level; the greater thedisorder the higher the entropy.What happens when entropy is 0?
The entropy of a closed system, determinedrelative to this zero point, is then the absolute entropy ofthat system. The entropy of a perfect crystal lattice asdefined by Nernst's theorem is zero provided that its ground stateis unique, because ln(1) = 0.Is the universe entropy?
The heat death of the universe, also known as theBig Chill or Big Freeze, is a conjecture on the ultimate fate ofthe universe, which suggests the universe wouldevolve to a state of no thermodynamic free energy and wouldtherefore be unable to sustain processes that increaseentropy.Why Does entropy increase with time?
Entropy (arrow of time) As one goes"forward" in time, the second law of thermodynamics says,the entropy of an isolated system can increase, butnot decrease. Hence, from one perspective, entropymeasurement is a way of distinguishing the past from thefuture.What is entropy in simple terms?
The entropy of an object is a measure of theamount of energy which is unavailable to do work. Entropy isalso a measure of the number of possible arrangements the atoms ina system can have. In this sense, entropy is a measure ofuncertainty or randomness.How much entropy is in the universe?
At the moment of the Big Bang, almost all of theentropy was due to radiation, and the total entropyof the Universe was S = 1088kB.Who came up with the second law of thermodynamics?
Around 1850 Rudolf Clausius and William Thomson (Kelvin)stated both the First Law - that total energy is conserved -and the Second Law of Thermodynamics. The Second Lawwas originally formulated in terms of the fact that heat does notspontaneously flow from a colder body to a hotter.Why is entropy a state function?
State functions do not depend on the path bywhich the system arrived at its present state. For example,internal energy, enthalpy, and entropy are statequantities because they describe quantitatively an equilibriumstate of a thermodynamic system, irrespective of how thesystem arrived in that state.Why do we need the second law of thermodynamics?
The second law of thermodynamics states that thetotal entropy of an isolated system can never decrease over time.In all processes that occur, including spontaneous processes, thetotal entropy of the system and its surroundings increases and theprocess is irreversible in the thermodynamicsense.Is the universe a closed system?
Closed systems cannot exchange matter with thesurroundings, but can exchange energy. Isolated systems canexchange neither matter nor energy with their surroundings, and assuch are only theoretical and do not exist in reality (except,possibly, the entire universe).Is an increase in entropy of a system sufficient to make a process spontaneous?
The second law of thermodynamics states that aprocess involving an isolated system will bespontaneous if the entropy of the systemincreases over time. In many processes, the increasein entropy of the surroundings is accomplished via heattransfer from the system to the surroundings (i.e. anexothermic process).What does Entropic mean?
In physics, an entropic force acting in a systemis an emergent phenomenon resulting from the entire system'sstatistical tendency to increase its entropy, rather than from aparticular underlying force on the atomic scale. Theentropic force can be considered as an emergent of theentropic interaction.What is the opposite of enthalpy?
The enthalpy of condensation (or heat ofcondensation) is by definition equal to the enthalpy ofvaporization with the opposite sign: enthalpy changesof vaporization are always positive (heat is absorbed by thesubstance), whereas enthalpy changes of condensation arealways negative (heat is released by the substance)What is Exergy analysis?
Exergy analysis is a powerful tool that allowsevaluating and analyzing processes by identifying theirimperfections due to thermodynamic irreversibilities.